
Where the MedTech Community Accelerates Regulatory Excellence
Expert guidance from regulators and industry to streamline compliance, stay deadline-ready, and advance patient safety through rigorous science
European Regulatory Affairs • Clinical Evaluations • SaMD & AI • Post Market Surveillance • Global Markets • Biocompatibility
ConnectMe Attendee Networking Platforn Now Open | Register Today to Start Scheduling 1-1s
Registered attendees can log into ConnectMe now to view the full attendee list, send meeting requests and build your personal agenda: https://lifesciences.connectmeinforma.com/
Book your seat today to access ConnectMe and secure the early bird savings rate. Save even more when you secure your hotel room at the JW Marriott Berlin with our attendee room block.
Your Path to MDR and IVDR Success: Overcome Today’s Challenges, Meet Tomorrow’s Deadlines, and Future-Proof Your Strategy
Gain insights to master the upcoming important revisions and stay prepared for deadlines. Achieve regulatory excellence and position your organisation as a leader in compliance and innovation.
Build meaningful connections with a community of top regulators and industry leaders. Participate in dynamic workshops and interactive roundtables, and take the first step in shaping your medical device regulation journey today.
Where MedTech Regulation Is Defined — By the People Behind It
With 80+ speakers already confirmed, the MedTech Summit 2026 is the must-attend event of the summer. Join notified bodies, competent authorities, and industry leaders from renowned organisations such as Johnson & Johnson, Philips, Roche, Baxter, and more. Gain actionable insights to streamline compliance, meet critical deadlines, and elevate patient safety through cutting-edge science
From Regulation to Innovation: What’s on the 2026 Agenda?
Stay ahead of the curve with expert insights designed to tackle today’s MedTech challenges and prepare for tomorrow’s opportunities. Gain the knowledge you need to innovate, comply, and lead in a rapidly evolving regulatory landscape.
European Regulatory Affairs
Overcome Bottlenecks, Stay Transition-Ready
Keep updated on proposed reforms to the EU MDR/IVDR and prepare for the EU AI Act and Digital Omnibus convergence. Navigate these upcoming major changes with confidence to remain compliant and competitive in 2026’s dynamic regulatory environment.
Software as a Medical Device & AI
Gain Strategic Insights in AI & Software Enabled Medical Devices
Discover cutting-edge market trends, tackling AI drift in PMS, explainable AI strategies, as well as emerging cybersecurity protocols. Gain the strategic foresight from key industry leaders, that is needed to innovate and adapt.
Biocompatibility
Ensure Safety, Avoid Compliance Risks
Explore the latest updates to ISO 10993-1 and master biocompatibility testing methods to ensure device safety and compliance. Avoid regulatory setbacks by gaining practical knowledge to streamline biological evaluations and meet global standards.
Global Markets
Expand Confidently Across Borders
Immerse yourself in the latest global regulatory updates and gain a comprehensive understanding of international requirements from the USA, Japan, China, LATAM, the UK, ASEAN, and GCC. Learn how to navigate global reliance efforts and ensure your products meet the demands of diverse markets without delays or compliance risks.
Clinical Evaluations & PMS
Streamline Processes, Stay Aligned
Optimise your clinical evaluation processes and align with current European and global post-market surveillance requirements. Avoid gaps in compliance and ensure your organisation is prepared to meet evolving expectations for safety and performance.
Workshop: SME Survival Guide
Actionable Strategies for SMEs
Gain practical insights and strategies tailored to SMEs navigating the rapidly evolving medical device regulatory sector. Learn how to overcome resource constraints, meet compliance requirements, and thrive in a competitive landscape.
Workshop: Biocompatibility
Hands-On Training for Compliance Excellence
Master biological evaluation and safety assessment for medical devices through interactive, hands-on training. Learn practical techniques to ensure compliance with ISO standards and maintain device safety, avoiding costly regulatory setbacks.
Who You'll Connect with at MedTech Summit
+
Competent Authority & Notified Body Speakers
+
Leading MedTech Industry Stakeholders
+
Medical Device, Software, and Combination Product Companies Represented
+
Countries Represented
Your Roadmap to Regulatory Excellence
Connect with regulatory, quality, clinical, and biocompatibility professionals, along with notified bodies, consultants, and future leaders, all shaping the future of MedTech. Explore tailored tracks like EU MDR, SaMD & AI, and Clinical & PMS, and gain expert insights, build connections, and overcome challenges with personalised programming. Ready to advance your career? Discover your path to success at MedTech Summit 2026.

Stay Ahead in Medical Devices Regulation. Anywhere, Anytime.
Unlock the MedTech Summit with the Digital Experience Pass. Access cutting-edge regulatory updates, expert insights, and vital networking opportunities—all in a fully digital format. Watch live-streamed sessions from Berlin, engage with speakers, and book online meetings with industry leaders via our exclusive ConnectMe platform.
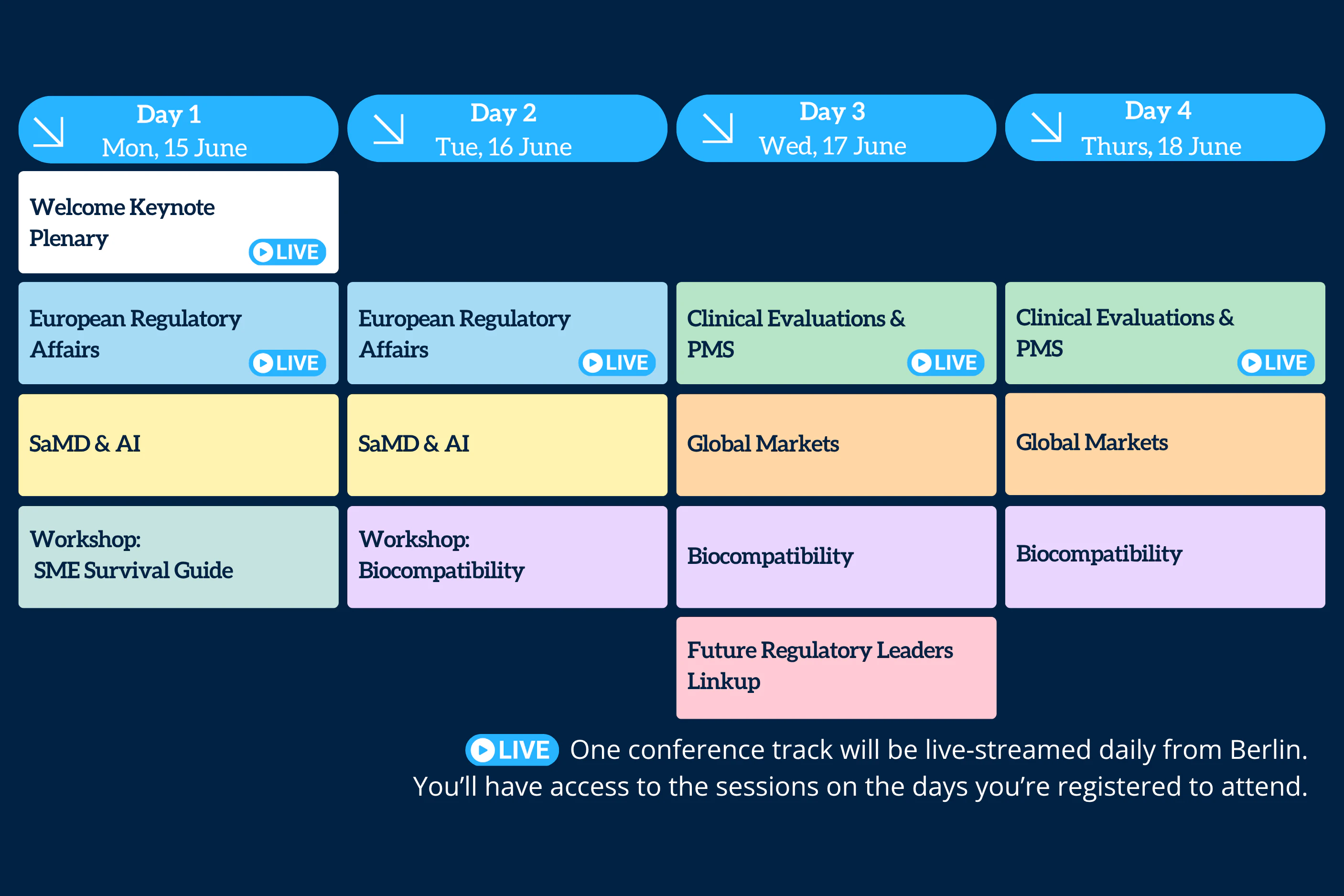
One track from the Berlin event will be live-streamed each day. View the full schedule here.
Engage Quality Decision-Makers at MedTech Summit!
Showcase your services to industry leaders and innovators. Let’s customise a sponsorship package that drives real results for your business.
Contact Neha Singh at Neha.Singh@informa.com
Stay in the Loop
FAQs
What is the MedTech Summit and who should attend?
The MedTech Summit is Europe’s leading conference on medical device regulation, bringing together regulatory affairs professionals, notified bodies, competent authorities, and industry leaders. It is designed for professionals involved in MDR/IVDR compliance, regulatory strategy, quality assurance, clinical affairs, and global market access.
When and where is the MedTech Summit taking place?
The MedTech Summit will take place from 15–18 June 2026 at the JW Marriott Hotel Berlin, Berlin, Germany.
How can I register for the MedTech Summit?
You can register directly on the website, where you’ll find ticket options, pricing, and additional event details.
Are there special pricing options for SME manufacturers?
Yes, the MedTech Summit offers a special SME rate for eligible applicants. To qualify, applicants must meet the following criteria:
- The SME must have fewer than 250 employees.
- The SME must be an active member of a medical device or in vitro diagnostic organisation with a device or diagnostic that is either already on the market or in the process of gaining certification.
- Applicants cannot be from a Competent Authority, Notified Body, Consultancy, or Vendor organisation.
- Applicants cannot be from an existing event sponsor or exhibitor organisation.
- Applicants cannot have a job title or focus in sales, marketing, account management, or business development.
What is the Digital Experience Pass, and who is it for?
The Digital Experience Pass allows attendees to participate in the MedTech Summit virtually, offering live-streamed sessions, on-demand content, and interactive features. It is ideal for professionals unable to attend in person but who want to benefit from the summit’s insights and networking opportunities.
What does the Digital Experience Pass include?
Each day, one track from the Berlin event will be live-streamed, allowing digital attendees to engage in real time and be part of the excitement. Also, the pass includes access to all session recordings, including those not live-streamed, on demand for up to 12 months after the event (subject to speaker permission).
Livestream Schedule:
- Day 1: Welcome Keynote Plenary & European Regulatory Affairs
- Day 2: European Regulatory Affairs
- Day 3: Clinical Evaluations & PMS
- Day 4: Clinical Evaluations & PMS
What are the key topics covered at the MedTech Summit?
The agenda addresses critical areas shaping the future of medical device regulation, including:
- European Regulatory Affairs (MDR & IVDR)
- Clinical Evaluations and Clinical Evidence
- Software as a Medical Device (SaMD) & Artificial Intelligence (AI)
- Post-Market Surveillance (PMS) and Vigilance
- Global Market Access and Regulatory Strategy
- Biocompatibility and Safety Requirements
Also, the agenda features keynote presentations, panel discussions, case studies, interactive roundtables, workshops, and deep-dive sessions.
Why is the MedTech Summit important for 2026?
With anticipated updates to EU MDR and ongoing IVDR transitions, 2026 is a pivotal year for regulatory compliance. The summit delivers direct insights from regulators and industry experts, equipping companies to navigate evolving requirements and mitigate compliance risks.
Will there be updates on EU MDR revisions at the summit?
Yes, dedicated sessions will cover expected EU MDR revisions, including implementation challenges, timelines, and strategic adaptations.








