63% of cell and gene therapy professionals changed their supply chain strategies due to COVID-19

89% of professionals working on cell and gene therapies say that the COVID-19 pandemic impacted their supply chain, while just under half say that the impact was significant, our recent industry report has found.
In fact, 63% of the survey respondents say that the pandemic had led them to change or re-evaluate their supply chain and manufacturing strategies.
Explore the full COVID-19 Response in Cell & Gene Therapy Manufacturing Report 2021 here.
The scramble for raw and ancillary materials
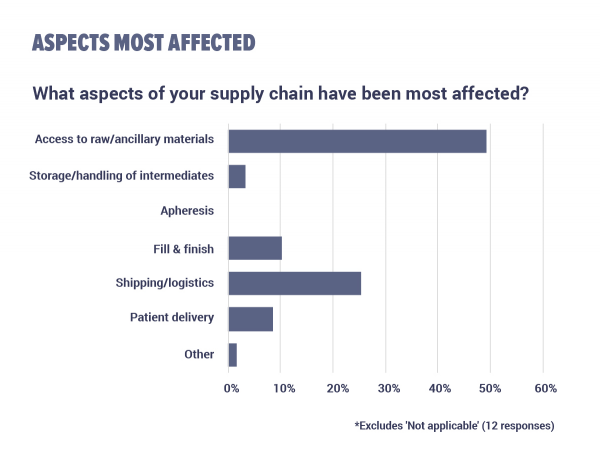
Access to raw and ancillary materials is the aspect of supply chains most affected by the COVID-19 pandemic, according to half of the survey respondents. Meanwhile, a quarter of respondents say that shipping and logistics in their supply chain took the worst hit.
Challenges in blood donation
Most of the respondents say that their blood donation and screening had been significantly affected by COVID-19, including process delays caused by extra care taken in donation, testing and collection.
The responses include:
- “Increased duration of collection due to additional screening and safety practices in place”
- “Significant reduction in donors”
- “Testing is must and that has delayed the process, incurring additional costs, and the worst issue is the donors are showing detest for the new procedure”
However, when asked which raw or ancillary material was the hardest to source, the respondents most commonly say that lab equipment such as PPE and single use consumables had been the most scarce. Biological supplies such as blood donations, antibodies and viral vectors followed closely behind.