The Leading Authority in
Pharmaceutical Compliance
Stay in front of enforcement priorities, navigate regulatory
complexity and drive greater impact across your compliance program.
With representation from CMS, DOJ, and SEC.
We're Celebrating PCC's 22-Year Legacy | Here's What's New!
We are thrilled to announce the confirmation of Centers for Medicare & Medicaid Services (CMS), Department of Justice (DOJ) and the United States Securities and Exchange Commission (SEC)!
PLUS:
+ All-Star Speakers & Counting
+ Hot-Topic Sessions
+ Interactive Networking Hours
+ CLE Credits*
*See CLE Credit Tab for More Information
Who are the industry-leading compliance and legal all-star speakers igniting the conversation at PCC 2026?
Join PCC 2026, the ultimate compliance conference where top pharma experts deliver actionable insights and strategies to drive compliance excellence and ethical leadership in the pharmaceutical industry!
How is PCC Gearing Up to Unleash Pharma Compliance Brilliance?
Whether you're a past attendee or a newcomer, get ready for an upgraded program packed with cutting-edge topics designed to supercharge your compliance program:
Address enforcement focus effectively in compliance programs
- Evaluate R&D pharma compliance risks under the False Claims Act
- Stay informed on the latest developments in diagnostic support programs
- Proactively address and mitigate commercial risks in patient support programs
- Strengthen your approach to managing social media challenges
Navigate regulatory uncertainty
- Examine ethical standards and enforcement drivers shaping Compliance programs
- Gain insights into enforcement risks through discussions with Department of Justice (DOJ) investigators
- Explore strategies for maintaining Compliance amidst unclear regulatory enforcement
- Navigate global programs effectively in the face of transformative enforcement changes
Unlock dynamic learning and unparalleled networking opportunities
- Tailored content: strategies designed for companies of every size and scope
- Dynamic formats: interactive use cases, roundtables, and actionable insights sessions
- Workforce empowerment: upskill your team and elevate digital literacy to drive success
- Build connections: icebreakers, community conversations and experience-based sessions
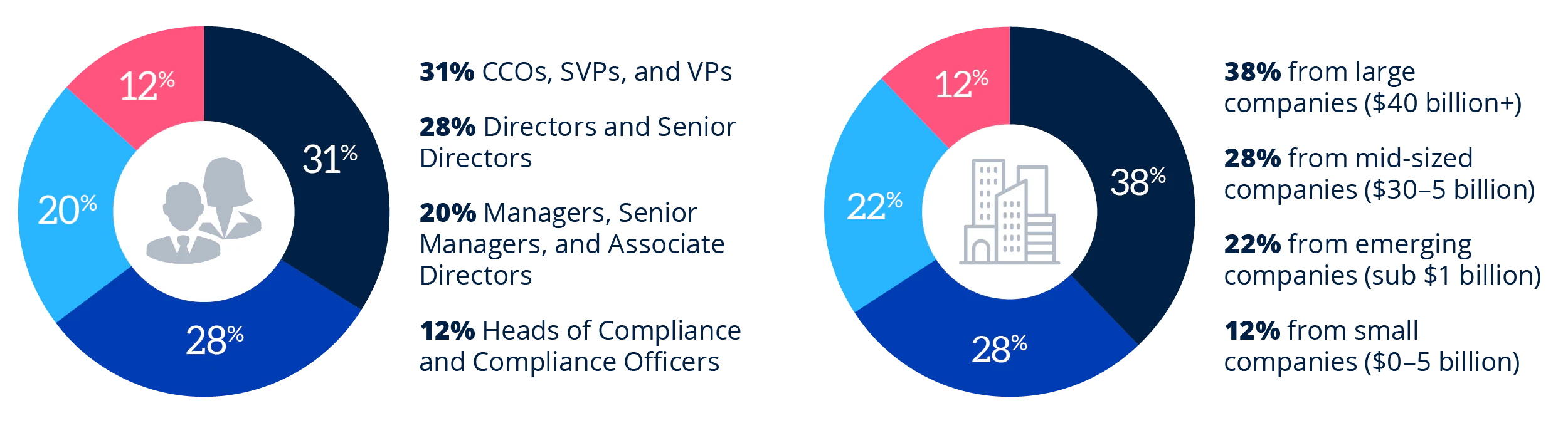
Who Will You Meet at the Pharmaceutical Compliance Congress?

Why Should You Join the Conversation at PCC 2026?
Feel the energy and ignite your curiosity as PCC takes center stage like never before! Prepare to be captivated by the insights of our remarkable Conference Chairperson, Lisa LeCointe-Cephas, Chief Compliance Officer at Johnson & Johnson Innovative Medicine.
Discover Lisa's unique perspective on the dynamic collaboration that drives groundbreaking innovation and fuels the spirit of progress.
Looking to Educate Your Entire Team at PCC 2026? New for 2026 | Rates as Low as $899 Per Person
Transform your compliance strategy with our most competitive team rates ever, from new starters to Senior Leaders, there is something for your entire organization. Maximize your professional development investment while ensuring your entire team gains critical industry insights. Secure your team's participation today by clicking below!
PCC Isn't Just Another Event, It's an Experience Designed to Expand your Network and Engage in Career-Advancing Conversations

The Pharmaceutical Compliance Congress Event Experience
Did you miss the 2025 event? Don't fret, here's a recap. Hundreds of your peers working within compliance, ethics, and legal functions united under one roof for electrifying discussions, groundbreaking industry benchmarking and unparalleled networking. Hit play to dive into the compliance conference event highlights and discover why this is the must-attend forum of 2026!
Connect and Network with Key Decision-Makers at Pharmaceutical Compliance Congress
Whether you’re looking to generate quality leads, showcase thought leaders or reinforce your brand, collaborate with us to identify custom sponsorship opportunities to help you reach your business objectives.
Contact Derek Tagliarino to learn more about sponsorship opportunities: DerekT@informaconnectls.com
Get the Latest Event Updates
FAQs
What is the Pharmaceutical Compliance Congress (PCC)?
PCC is the premier life sciences compliance and legal event that brings together senior leaders, compliance professionals and legal experts to explore enforcement trends, strategic compliance practices and regulatory frameworks shaping the pharmaceutical industry.
When and where is PCC 2026 taking place?
PCC 2026 will take place on April 27-29, 2026 at The Ritz-Carlton, Tysons Corner in McLean, Virginia, USA.
Who attends PCC?
This congress is ideal for:
- Senior compliance officers and C-suite executives looking to gain strategic insight and connect with peers
- Mid-level and rising compliance professionals seeking practical skills, enforcement trends, and networking opportunities.
- Legal counsel, risk and ethics professionals, regulatory leaders, and other experts within the pharmaceutical, biotech, and healthcare sectors aiming to strengthen and future-proof their compliance programs
What will I learn at PCC?
At PCC, you will gain practical knowledge and strategic insights to strengthen and future-proof your compliance programs. Key learning outcomes include:
- Regulatory readiness: Learn strategies to anticipate and adapt to regulatory and enforcement changes across healthcare and life sciences.
- Compliance resilience: Understand how to build strong, agile compliance teams that can respond effectively to risk and disruption.
- Data-driven compliance: Explore how data analytics can be used to improve monitoring, risk detection, and decision-making.
- Enforcement insights: Hear directly from representatives of federal agencies such as the DOJ, FDA, and CMS, along with perspectives from prosecutors, regulators, and leading law firms on current enforcement trends and priorities.
- Organizational impact: Learn how compliance programs create enterprise value, support ethical culture, and drive operational excellence.
- Global compliance alignment: Examine approaches to harmonizing compliance standards across regions and regulatory regimes.
- Risk management in emerging areas: Address evolving risks related to commercial practices, social media engagement, and patient support programs.
- Turning challenges into opportunities: Discover how organizations can use regulatory and enforcement challenges to improve culture, governance, and operations.
By the end of the program, participants will be equipped with actionable tools and insights to navigate complex and evolving regulatory landscapes with confidence.
Can I network and benchmark with industry peers?
Definitely! Pharmaceutical Compliance Congress (PCC) features dedicated networking opportunities, interactive sessions, community conversations, and CCO benchmarking discussions designed to foster collaboration and expand professional networks.
Who are the featured speakers at PCC 2026?
Our agenda includes senior compliance officers from top pharmaceutical companies, U.S. Attorneys, legal experts and thought leaders such as:
How do I book my place at the Pharmaceutical Compliance Congress (PCC)?
Book your pass for PCC on our official 'Book Now' page here or request information about our group rates here.
Does PCC provide continuing education or professional credits?
This event qualifies for CLE credits. CLE credits will only be awarded to sessions watched live at the conference. Sessions watched On-Demand at a later date are not eligible for CLE credits.
To qualify for CLE credits as an in-person attendee, you must pick up your conference badge with Informa staff upon your arrival at the registration desk and wear your badge for the entirety of the conference. Credit calculations will be conducted according to your onsite participation data collected through Crowd Connected.
Please see our CLE Credits page for more information.
Who organizes PCC?
PCC is organized by Informa Connect, take a look at the wider biotech/pharma events that Informa runs here.








